Twenty percent of Earth's oxygen is produced by the Amazon forest.
Wow, I wonder how many % of oxygen is produced by Sabah's forest??!
The noble gas Xenon lasers can cut through materials that are so tough even diamond tipped blades will not cut.
Diamond is the hardest element man.. amazing huh..
Gold and Copper are the only two non-white metals.
really????
The burning sensation we get from chilli peppers is because of a chemical called Capsaicin.
hmmm... so this capsaicin is detected by all our taste buds ka?
The lighter was invented before the match (in 1816 by J.W. Dobereiner).
huh? Really.. so y v still need match when v have lighter???
Each time lightning strikes, some Ozone gas is produced, thus strengthening the Ozone Layer in the Earth's atmosphere.
God... send some lightning & thunder...
Honey does not spoil.
God's creation... x expiry date... wonderful...
There's enough gold in the Earth's crust to cover the entire land surface knee-deep.
I wonder where's all the gold now hahaha....
An average adult body contains around 250g (1/2lb) of salt.
ewwww... that's a lot!!!
Followers
Friday, January 30, 2009
Thursday, January 22, 2009
what 2 do IF u r failing to understand ur chemistry???
I suspect I don't have to tell you this: not everyone who takes chemistry passes it. If you're taking a chemistry course and it feels like a sliding down into a deep dark hole to your doom, you may be failing. Sometimes you can turn it around. Sometimes you can't. Either way, it's good to have a plan.
What Not to Do?
First, let's take a look at how not to handle the situation. You may view failing chemistry as the end of the world, but how you react could actually make a bad situation worse, so don't do these things: Panic /Cheat /Threaten your teacher/ Attempt to bribe your teacher/ Give up/ Do nothing...
Steps to Take:
Talk to Your teacher or any chemistry teacher/ Keep doing your homework/ keep attending ur chemistry class/ take notes/ get someone's note/ try a different text/ work problems...
Failure isn't the end of the world, which is good, since everyone experiences failure at some point or another. Even if you fail the class, if you learned how to face failure successfully then it wasn't a total waste.
Wednesday, January 21, 2009
Chemical Formulae & Equations
Mole= Mass/RAM or RMM
Number of particles= mole X Avogadro Constant
(remember, number of atoms, ions and molecules must be identified as the calculation varies)
Example, Number of particles for 1 mole of H2 is 6.02 X 1023 molecules BUT Number of atoms will be 2 X 6.02 X 1023 ( one hydrogen molecule has 2 hydrogen atoms!)
Mass of a substance = Mole X Molar Mass
(Molar mass is calculated as RAM or RMM BUT the Unit is g per mol)
Volume of Gas = Mole X Molar Volume
(For STP, molar volume is 22.4 whereas, for room conditions, molar volume is 24)
X NA X Molar Mass
number of particles ----> mole ----> mass
l
l X Molar Volume
v
Volume of gas
Notes: 1 dm3 = 1000 cm3
Number of particles= mole X Avogadro Constant
(remember, number of atoms, ions and molecules must be identified as the calculation varies)
Example, Number of particles for 1 mole of H2 is 6.02 X 1023 molecules BUT Number of atoms will be 2 X 6.02 X 1023 ( one hydrogen molecule has 2 hydrogen atoms!)
Mass of a substance = Mole X Molar Mass
(Molar mass is calculated as RAM or RMM BUT the Unit is g per mol)
Volume of Gas = Mole X Molar Volume
(For STP, molar volume is 22.4 whereas, for room conditions, molar volume is 24)
X NA X Molar Mass
number of particles ----> mole ----> mass
l
l X Molar Volume
v
Volume of gas
Notes: 1 dm3 = 1000 cm3
Wednesday, January 14, 2009
Upcoming! Trip to KK - Feb 2009
Boys & Girls,
Teacher Donny & I had planned a Trip for all of u to KK on February 12-13.
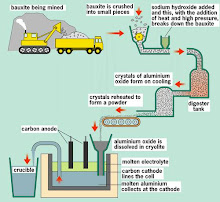
We will be visiting Electroplating & Aluminium Factory, Milk processing Factory,
Formaldehide Factory, and also Noodle factory.. all these r kind of related to your chemistry, physics and biology... accompanying teachers will be Mr. Robert, Mr. Donny, Madam Norhafiza & myself. you'll need to pay RM50 for this trip. We'll be staying at SaBah Kinabalu Hostels, Likas. Please spread the news.. formal letter will be given out soon.. probably by next week ya
Teacher Donny & I had planned a Trip for all of u to KK on February 12-13.
We will be visiting Electroplating & Aluminium Factory, Milk processing Factory,
Formaldehide Factory, and also Noodle factory.. all these r kind of related to your chemistry, physics and biology... accompanying teachers will be Mr. Robert, Mr. Donny, Madam Norhafiza & myself. you'll need to pay RM50 for this trip. We'll be staying at SaBah Kinabalu Hostels, Likas. Please spread the news.. formal letter will be given out soon.. probably by next week ya
Atomic Structure
Things you must master here are:
1. Kinetic theory of gas & matter (solid, liquid & gas)
2. Heating & Cooling graph
3. Nucleon No, Proton No & Neutron No
4. Electron arrangement/ electron configuration of an element
Any problem understanding these subtopics? Post ur opinion here. U can write
down tips n discuss ur problem....
1. Kinetic theory of gas & matter (solid, liquid & gas)
2. Heating & Cooling graph
3. Nucleon No, Proton No & Neutron No
4. Electron arrangement/ electron configuration of an element
Any problem understanding these subtopics? Post ur opinion here. U can write
down tips n discuss ur problem....
Saturday, January 10, 2009
How is Chemistry class so far?
Boys and Girls, How do you find our lesson so far? Any problem that you would like to share here? Please feel free to drop in a few comments. Thanks.
Subscribe to:
Posts (Atom)